Model Of Atom By Ernest Rutherford
Model Of Atom By Ernest Rutherford. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …
Prezentováno List Of The Atomic Theories In Order Dalton S Atomic Theory
The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.
It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford... It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself... The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve... It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.
14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called ….. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called ….. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.

The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic... .. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons... The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself... In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons... In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve... The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.
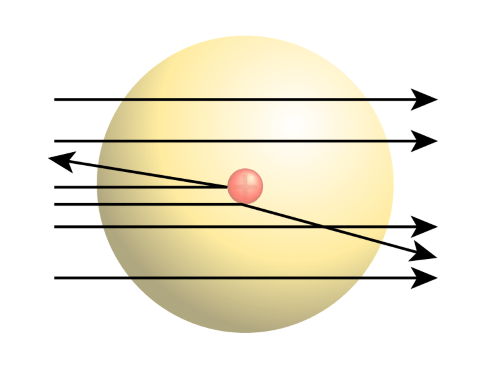
The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. . The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic... In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford... . In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. . The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons... It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. . The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.

The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.
In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called ….. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called ….. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself... The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself... The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic... In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …
It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve... The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic... It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself... The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. . The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve... The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself... The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.
It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself... The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford... The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called ….. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons... 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.
The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic... It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. .. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called ….. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve... In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. .. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve... The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. .. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …
In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself... The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.

The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself.. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.

14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself... The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …

The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve... The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons.. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.

It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve... The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.

In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself... The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called …
In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.
It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve... 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford. In 1909 the geiger and marsden experiment was performed, also known as the rutherford experiment, as it was led by rutherford himself. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve. It describes the atomic model as to where all the atom's mass is concentrated in the centre called the nucleus, around which the negative charges called the electrons revolve.
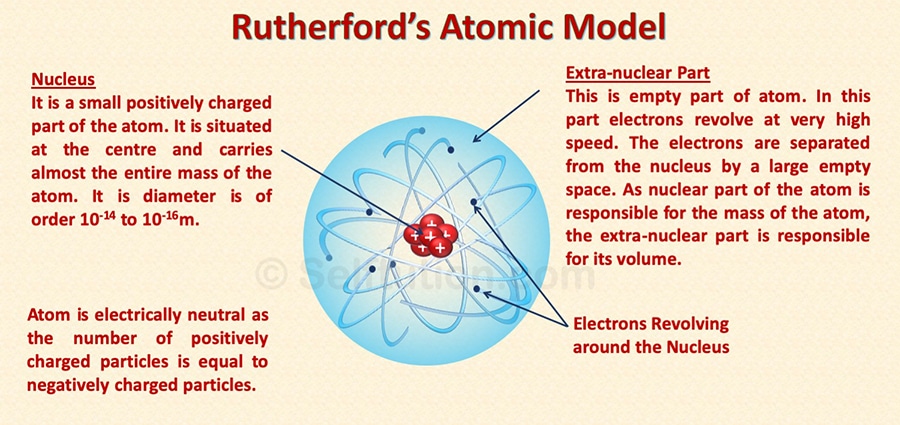
The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons... 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.

The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic. The model described the atom as a tiny, dense, positively charged core called a nucleus, in which nearly all the mass is concentrated, around which the light, negative constituents, called … The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. 14.08.2021 · rutherford's atomic model or planetary model of the atom is a model proposed by ernest rutherford.. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.

The rutherford atomic theory has defined the atom as a tiny, dense, positively charged core called a nucleus, which is surrounded by negative charges called electrons. The rutherford scattering observed in the investigation suggested that the early panettone and saturnian atomic.